Invented by KIM; Young-Ki, CHOI; Aram, KIM; Sangmi, DOO; Sungwook, KANG; Gwiwoon, LEE; Soonrewl

Let’s take a deep dive into a new patent application for a positive electrode active material in lithium batteries. We’ll walk through what makes this invention special, why it matters, and how it stacks up against what’s already out there. By the end, you’ll know what this technology brings to the table and why it could be a big deal for the future of batteries.
Background and Market Context
Today, everything from your phone to electric cars runs on batteries. The world wants batteries that last longer, charge faster, and are affordable. Electric vehicles, for instance, need batteries that can hold a lot of energy without taking up too much space or costing a fortune. Phones and laptops demand batteries that are safe, can be recharged many times, and won’t wear out quickly. As these devices become more common, the push for better batteries gets stronger.
Right now, most rechargeable batteries use lithium ion technology. These batteries work by moving lithium ions between a positive electrode (the cathode) and a negative electrode (the anode). The positive electrode is where a lot of the action happens. It needs to hold a lot of lithium, must be stable for many charge cycles, and should not cost too much to make. If we can improve the materials in the positive electrode, we can make batteries that last longer, are safer, and are more powerful.
But there are challenges. Some materials work well but are very expensive or use metals like cobalt, which is costly and comes with sourcing concerns. Others are safe and cheap but can’t store as much energy. Finding the right mix of materials is tricky. Makers of phones, laptops, and electric vehicles all want batteries that meet their needs for cost, safety, and performance. As a result, there is a race to invent new positive electrode materials that hit the sweet spot between power, price, and safety.
This invention seeks to address these needs by offering a new positive electrode active material. It combines different types of particles, each with its own structure and chemistry, to create a better electrode for lithium batteries. The goal: higher energy density, longer life, and lower cost, all rolled into one.
Scientific Rationale and Prior Art
To understand why this new material matters, let’s look at the science and what others have tried before.
In current batteries, the most common positive electrode materials are:
- Lithium cobalt oxide (LCO): Offers high energy, but uses expensive and less stable cobalt.
- Lithium iron phosphate (LFP): Very safe and stable, but stores less energy compared to LCO. It has an “olivine” structure, which makes it sturdy but limits its voltage and energy output.
- Lithium manganese oxide (LMO): Uses the “spinel” structure. It’s cheaper and has good power output but can have stability issues over time.
- Nickel-rich layered oxides (like NCM or NCA): High energy density and power, but more expensive and less stable under stress.
Many researchers have tried mixing these materials or tweaking them with other elements to get the best of all worlds. For instance, adding manganese to LFP can boost voltage, while adding magnesium or aluminum can stabilize the structure. Coatings of carbon or other compounds can help particles last longer and conduct electricity better. Some have tried using “sacrificial” materials that give up lithium in early cycles to help stabilize the battery, but these often add cost or complexity.
Despite all this work, challenges remain. If you use too much of a cheap material like iron phosphate, the battery is safe but not powerful. If you use too much of a high-energy material, the battery may wear out faster or become less safe. Mixing different types of structures (olivine, spinel, and layered) is tricky, as they react differently during charge and discharge.
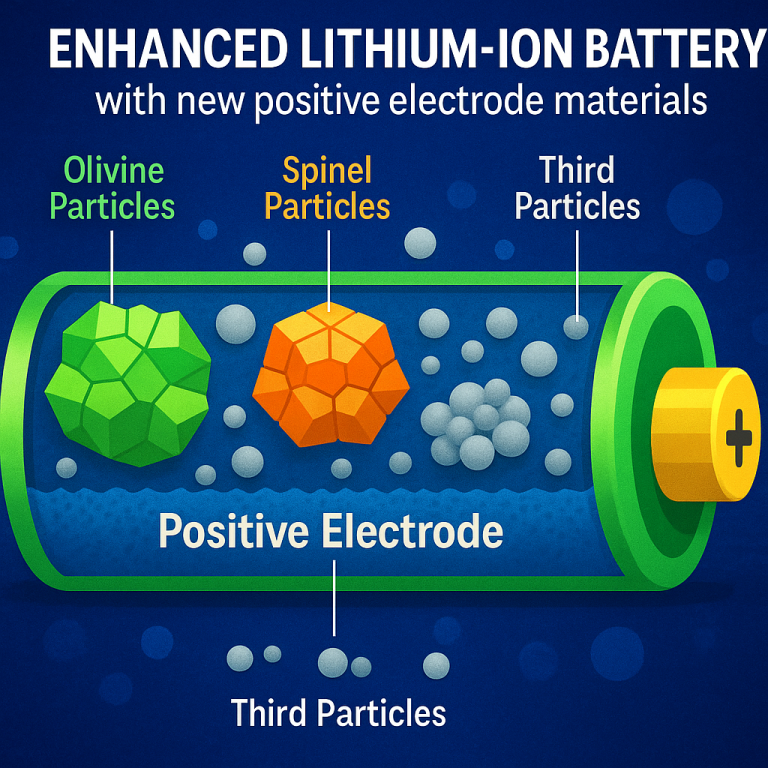
What’s new here is the careful balance of three main types of particles:
Olivine-type particles: These are made from a lithium iron phosphate base, tweaked with small amounts of other metals (like titanium, magnesium, vanadium, or niobium) to keep the particles small and uniform. This helps the battery last longer and work better at low temperatures.
Spinel-type particles: Based on lithium manganese oxide, they are doped with magnesium or aluminum to keep them stable. The spinel structure lets lithium ions move quickly, giving good power output.
Sacrificial particles: These are rich in lithium and iron, like Li5FeO4. They give up extra lithium during the first charge, making up for losses that happen when the battery forms its protective layers (called the SEI). After that, they become inactive and don’t take part in further battery cycles.
What’s clever is the way these are mixed: the olivine and spinel particles make up the bulk of the electrode, while just a small amount of sacrificial material is added—enough to help, but not so much that it hurts performance or safety. The patent also talks about adding a fourth type of particle (like nickel-rich layered oxides) for even more energy if needed.
This approach builds on prior work, but refines it. By controlling particle size, using careful coatings, and getting the ratios just right, this invention aims to provide a positive electrode material that is cheap, powerful, long-lasting, and safe. The patent’s data shows the new mixture has better energy density and longer life than older materials.
Invention Description and Key Innovations
Let’s walk through what’s actually in this invention and why it matters.
The core idea is to make a positive electrode material with three different kinds of particles:
First particles: These are based on lithium iron phosphate (LFP), with a crystal structure called olivine. Small amounts of titanium, magnesium, vanadium, or niobium are added to keep the particles small and even, which helps the battery work better and last longer. These particles can either be single, tiny crystals or made up of lots of smaller ones stuck together. They are often coated with carbon to help conduct electricity. Sizes can range from about 200 nanometers (nm) to several micrometers (μm), and the coating helps them stay stable during charging and discharging.
Second particles: These are lithium manganese oxide (LMO) with a spinel structure. They are doped with magnesium or aluminum. The spinel structure allows lithium ions to move in and out easily, which gives the battery good power. These particles are also made up of smaller primary particles clumped together. The size and uniformity are carefully controlled, and sometimes an extra coating is added to keep them from breaking down over time.
Third particles: These are lithium-rich iron oxides, like Li5FeO4. Their main role is to release “extra” lithium during the first battery charge. This helps the battery make up for lithium lost when forming the solid electrolyte interface (SEI) on the negative electrode. Without these sacrificial particles, batteries lose capacity over time. Only a small amount (between 0.5 and 1.5 parts per 100 parts of the total electrode material) is added—enough to help, but not so much that it creates safety or design problems.
Sometimes, a fourth particle type is added, like a nickel-rich oxide with a layered structure, for even more energy. But the three core types are what set this invention apart.
The patent also lays out how to make these particles and how to mix them. It gives detailed steps for preparing each particle type, such as mixing, grinding, drying, and baking, as well as how to combine them into an electrode. The patent’s claims cover things like particle size, mixing ratios, types of coatings, and the exact chemical formulas used.
Here are the key innovations:
Smart particle mix: By combining olivine and spinel particles in a careful ratio (usually between 4:6 and 6:4 by weight), the invention balances safety, cost, and energy output. The manganese content is kept in a sweet spot (about 50-60% of the metal content) to get the best of both worlds: good voltage from manganese and good stability from iron.
Use of sacrificial particles: Adding a small amount of lithium-rich iron oxide helps make up for the lithium lost during the first battery charge. This keeps the battery’s capacity high and helps it last longer. Once the lithium is released, these particles become inactive and don’t interfere with later battery cycles.
Controlled particle size and coatings: By making sure all the particles are the right size and have the right coatings, the electrode is more stable and conducts electricity better. The carbon coating on the olivine particles, for example, helps the battery work better at higher currents and keeps the particles from breaking down.
Flexible design: The approach allows for adding other types of particles (like nickel-rich oxides) to push energy density even higher. The patent is careful to specify ranges for all the important features—like particle size, mixing ratios, carbon content—so that the material can be customized for different types of batteries or uses.
Proven results: The patent provides data showing that batteries made with this new material have higher energy density (over 450 Wh/kg), excellent capacity retention (about 98.5% after 50 cycles), and good safety. The mixture also has a higher pellet density, which means more active material can be packed into each battery cell.
In practice, this means batteries with this electrode material can be smaller, last longer, and be made more cheaply. This matters for electric cars (which need long range and safety), phones (which need long life and fast charging), and many other devices. Because the approach uses less cobalt and more common metals like iron and manganese, it should also be more sustainable and less subject to price swings.
The patent even covers the details of how to process the electrode: how to mix the powders, what binders and conductive materials to use, and how to make the electrode layer on an aluminum collector. This makes it easier for manufacturers to adopt the technology.
Conclusion
The positive electrode active material described in this patent is a smart step forward for lithium batteries. By mixing olivine, spinel, and sacrificial particles, all with careful control of size, structure, and chemistry, the invention delivers a material that is safe, powerful, and cost-effective. The design lets battery makers use less expensive metals, pack more energy into each cell, and keep batteries working well over many charge cycles. Real-world data in the patent shows this approach works, offering higher energy density and longer life than older materials.
If you are in the battery industry, or if you rely on devices powered by lithium batteries, this invention is worth watching. It has the potential to make batteries cheaper, safer, and more powerful—something we all want as our world becomes more dependent on rechargeable power. By carefully balancing the best features of different materials and solving old problems like lithium loss, this technology could help power the next generation of phones, cars, and beyond.
Click here https://ppubs.uspto.gov/pubwebapp/ and search 20250336927.
