Invented by KIM; Young-Ki, CHOI; Aram, KIM; Sangmi, DOO; Sungwook, KANG; Gwiwoon, LEE; SOONREWL
New ideas are changing how we build and use batteries. This patent application introduces a fresh way to make the positive electrode inside rechargeable lithium batteries, aiming to make them last longer, hold more energy, and cost less. Let’s dive into the details and see what makes this invention stand out.

Background and Market Context
Rechargeable lithium batteries are everywhere. You will find them in your phone, laptop, tablet, and even in electric cars. As the world wants cleaner energy and gadgets that last longer, the need for better batteries grows every year. Companies and researchers are always looking for ways to make batteries work harder, last longer, and cost less.
In the past, battery makers used materials like lithium cobalt oxide, lithium iron phosphate, or lithium manganese oxide for the positive electrode. These materials help the battery store and release energy safely and quickly. But each one has its own problems. Some are expensive, like cobalt. Some do not hold enough energy, while others do not last long or are not safe under some conditions.
As people use more electric cars and big energy storage units, batteries must become even better. They need to be safe, powerful, cheap, and last a long time. This is not easy! Companies want to use less cobalt because it is expensive and sometimes hard to get. They also want batteries that do not overheat or wear out too quickly. So, the race is on to find new materials and ways to put them together inside the battery.
The main part of this patent is about the positive electrode, also called the cathode. This is the part of the battery where lithium ions move in and out during charging and using the battery. If the positive electrode is made better, the battery can be safer, last longer, and store more energy. That’s the challenge this invention tries to solve—making a positive electrode that is strong, stable, less costly, and easy to make.
Scientific Rationale and Prior Art
To see why this invention matters, let’s look at how batteries work and what has already been tried. Lithium batteries have two main parts inside: a positive electrode (cathode) and a negative electrode (anode), with a separator and a special liquid called the electrolyte. When you charge or use the battery, lithium ions move back and forth between the electrodes. The way these parts are made and what they are made from decides how good the battery is.
For the positive electrode, scientists have used materials like:
- Lithium cobalt oxide (LCO): Holds lots of energy but uses a lot of cobalt, which is expensive and sometimes comes from places with supply problems.
- Lithium iron phosphate (LFP): Safer and lasts longer but holds less energy.
- Lithium manganese oxide (LMO): Safer and cheaper but does not last as long.

Each of these has strengths and weaknesses. For example, LCO is great for phones but too costly for big batteries in cars. LFP is very safe, but your phone would not last as long on one charge. LMO is fairly cheap and safe but breaks down faster after many charges.
Other scientists have tried mixing these materials or changing their shapes. Some make very small particles (nano-sized), add coatings to protect them, or mix in other metals (called doping) to help them work better. Sometimes, they put two or more materials together in layers or blends, hoping to get the best of both worlds. For example, mixing spinel (like LMO) with olivine (like LFP) can sometimes help batteries last longer and work at higher voltages, but it’s tricky to get the mix just right.
Still, there have been problems:
- Mixed materials sometimes separate or react badly with each other.
- Some designs need lots of binder (a glue-like material), which lowers the energy the battery can store.
- Particle shapes and sizes can make it hard to pack the electrode tightly, wasting space.
So, the search continues for a way to put these materials together so they help each other, not hurt each other. The goal is to use less binder, make the particles stick well to each other and to the current collector, and keep the battery working well even after many charges.
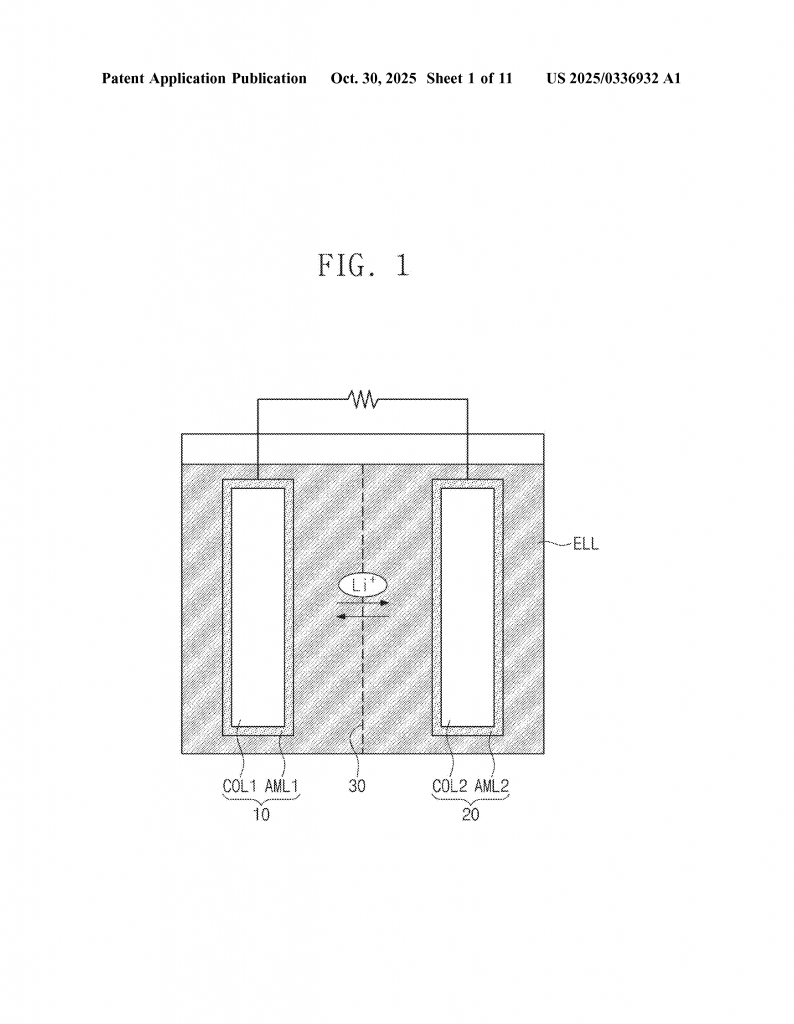
Invention Description and Key Innovations
This patent application offers a new plan for building the positive electrode in rechargeable lithium batteries. The inventors use a double-layer structure with carefully chosen materials and particle shapes. Let’s break it down step by step.
The Double-Layer Structure
Instead of mixing all the active materials together in one layer, the inventors stack two layers, each with different kinds of particles:
- First Layer (closest to the current collector): This layer has two types of particles. One is made from an olivine-structured compound (like lithium manganese iron phosphate) shaped as single, small particles. The other is a spinel-structured compound (like lithium manganese oxide) shaped as larger, secondary particles made from many small crystals stuck together.
- Second Layer (on top of the first): This layer has another olivine-type material, but here the particles are bigger secondary ones, each made from bunches of tiny primary particles.
This design is smart for several reasons. The layer next to the current collector uses smaller particles and spinel-type material, which helps electrons and lithium ions move quickly. The second layer uses bigger, porous particles that help the electrode hold more energy and let lithium ions move in and out easily. By stacking them, the battery gets the power and speed from the first layer, and the energy storage and stability from the second.
Why These Particles and Structures?
Each particle type is chosen for a reason:
- Olivine-structure (like LFP or lithium manganese iron phosphate): Known for stability and safety. Single, small particles offer fast lithium movement, while secondary particles (made of many tiny crystals) offer high energy storage and good stability.
- Spinel-structure (like LMO): Known for high voltage and fast charging, but usually does not last as long alone. By blending it in the first layer, it gives the electrode a voltage boost and helps with conductivity.
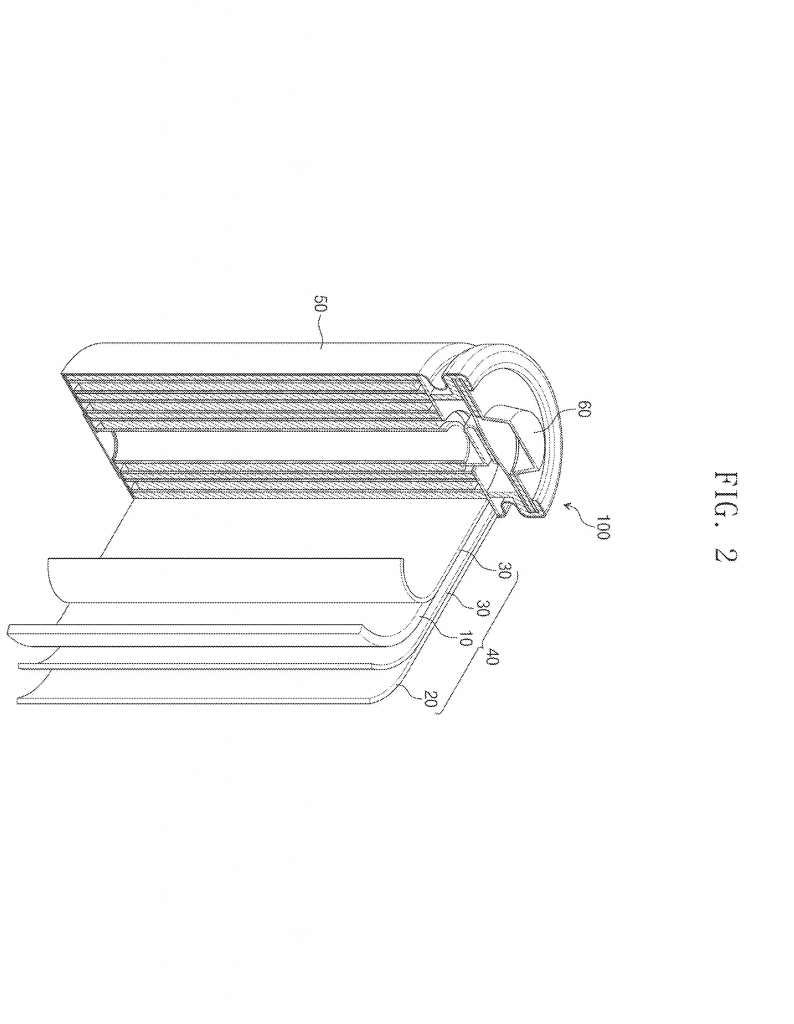
The inventors also control the particle size very carefully. Smaller particles in the first layer help make a good connection with the current collector. Larger, porous particles in the second layer offer a good path for lithium ions and help the electrode hold together. The right ratio of these materials, by weight, is also important. The spinel-type material is kept at about 15–30% by weight to balance voltage and lifetime.
Special Additives and Binders
Another part of the invention is about how the layers are made. Each layer gets a “functional additive”—a mix of binder (which holds the particles together) and conductive material (which helps electricity flow). The first, bottom layer uses less binder and conductive additive. This is possible because the mix of small and large particles helps the layer stick to the current collector with less glue. Using less binder leaves more room for active material, which means more energy can be stored.
The second, top layer uses more binder and conductive additive. Here, it is needed to hold the bigger, porous particles together and keep the layer strong over many charge cycles. The inventors control the ratio between the two layers, so the binder and additive use is just right—not too much, not too little.
Porosity and Particle Size Control
The secondary particles in the top layer are made to be porous, with lots of tiny openings inside. This makes it easier for lithium ions to move in and out, helping the battery to charge fast and work well at low temperatures. The inventors also watch the “span” (a measure of how evenly sized the particles are), keeping it between 0.3 and 0.75. Particles that are all about the same size pack together better, making the electrode stronger and more efficient.
Manganese Doping
Manganese is added in controlled amounts to improve the battery’s average voltage and lifetime. The inventors calculate just how much manganese to add, keeping the total in the electrode between about 0.50 and 0.60. Too much or too little can hurt the battery’s performance, so this careful balancing act is key.
Improved Performance in Real Batteries
The inventors built batteries using this new positive electrode and tested them. Compared to older designs, the new batteries have:
- Higher energy density (more power stored for the same size and weight)
- Better voltage (the battery can output more power)
- Improved cycle life (the battery lasts longer even after many charges)
- Lower electrical resistance (faster charging and less waste heat)
- Good performance at low temperatures (useful for cars in cold places)
They also found that making the electrode with two layers, instead of one mixed layer, uses less binder overall. That means more room for active material, so the battery holds more energy. The double-layer design also makes the electrode easier to manufacture, stick to the current collector, and remain strong over time.
How to Make the Electrode
The patent also describes how to actually make these particles. The inventors use known methods like mixing powders, grinding, spray drying, and careful heating in a special oven. By controlling the recipe and how the particles are treated, they get the right size, shape, and structure for each layer. After making the powders, they mix each with the right amount of binder and conductive material, then coat the first layer onto a metal (often aluminum) current collector. After drying, the second layer is coated on top. Finally, the electrode is pressed flat and baked again to finish.
Applications and Benefits
This new double-layer positive electrode can be used in almost any lithium battery. It is good for phones, laptops, power tools, and especially electric cars and big batteries for storing energy from solar or wind. The design is safe, uses less expensive cobalt, works at higher voltages, and is easier to make in big factories. Battery makers can use less binder, which means better energy storage and lower cost. The batteries also last longer, even when charged and used many times.
Conclusion
The world needs better, safer, and more powerful batteries. This patent gives a new way to build the positive electrode, using two special layers with different particles. By mixing olivine and spinel structures, picking the right particle sizes, and stacking them just right, the inventors create a battery that can store more energy, last longer, and cost less to make. This double-layer electrode is a strong step forward for all kinds of rechargeable lithium batteries, from your phone to your car. By understanding the science and smart design behind this invention, battery makers and users alike can look forward to better performance and more reliable power in the future.
Click here https://ppubs.uspto.gov/pubwebapp/ and search 20250336932.
