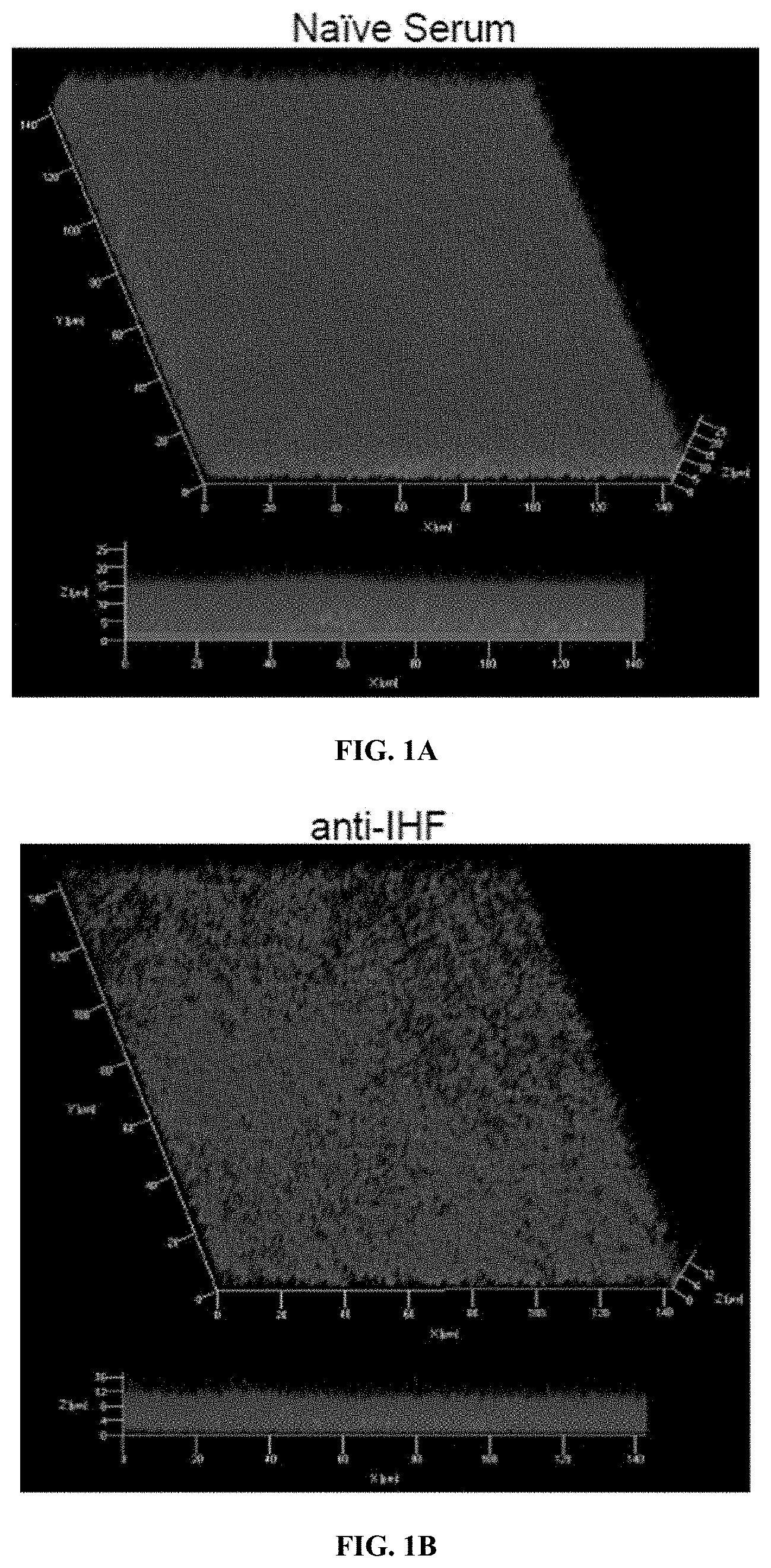
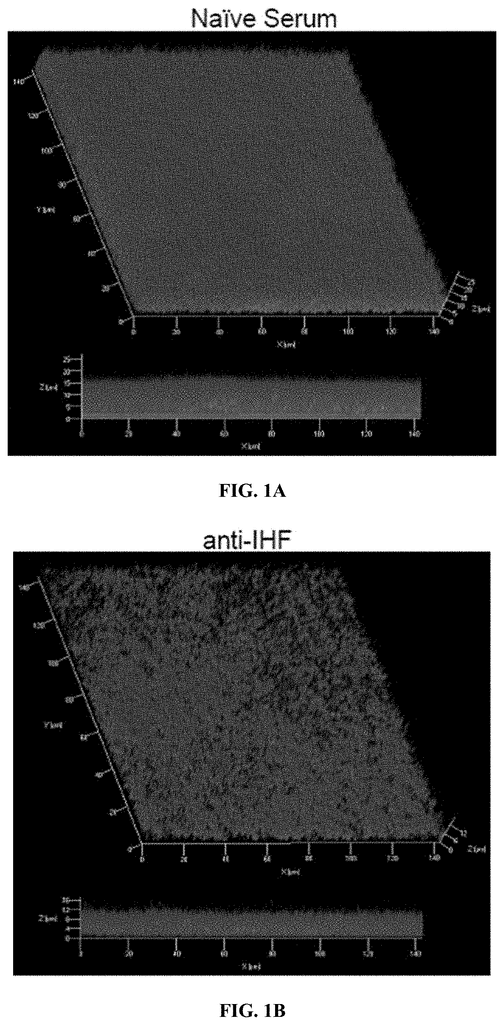
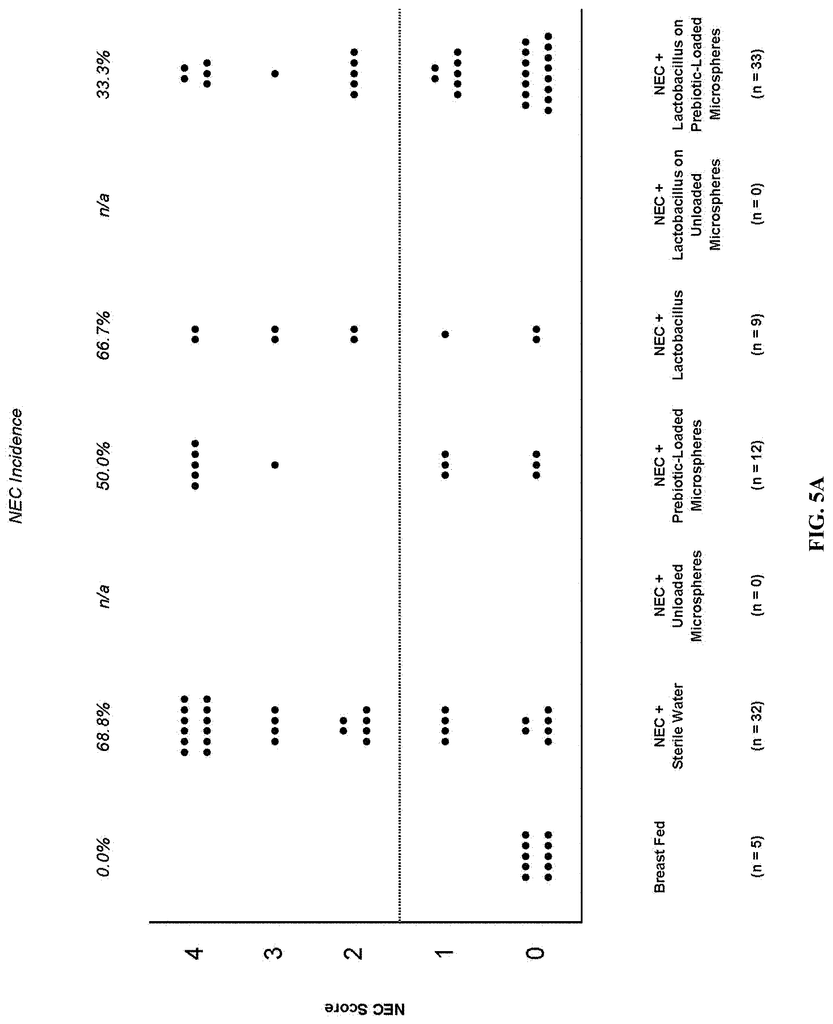
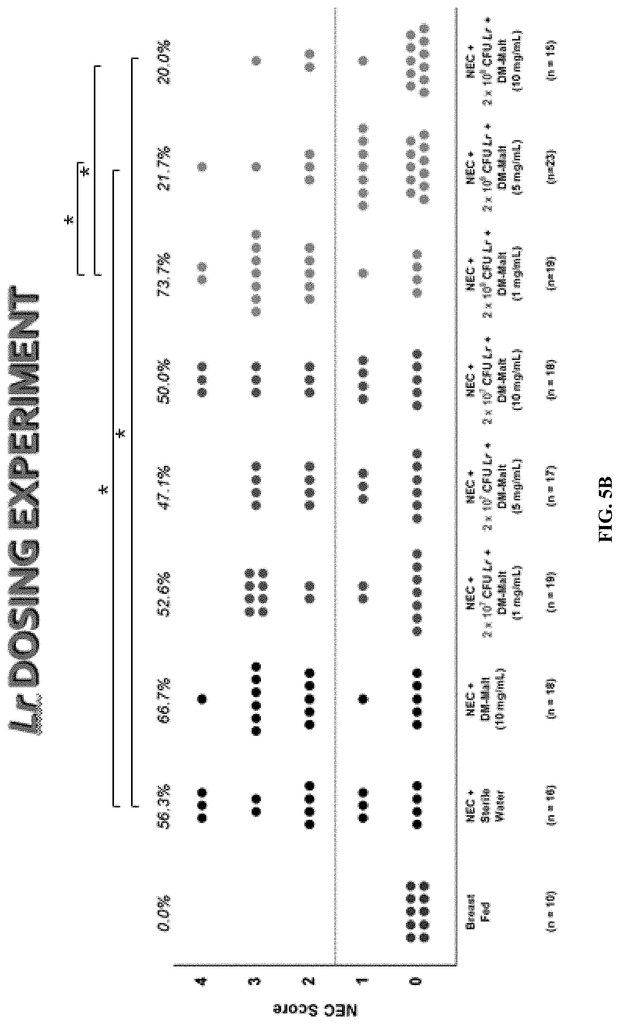
Invented by Steven D. Goodman, Lauren O. Bakaletz, Michael Bailey, Gail Besner, Ohio State Innovation Foundation
The demand for prebiotic formulations has been driven by various factors, including an increasing interest in natural and functional foods, rising awareness about the importance of a balanced gut microbiome, and a growing number of people suffering from digestive disorders. Additionally, the COVID-19 pandemic has further highlighted the importance of maintaining a strong immune system, which is closely linked to gut health.
One of the key drivers of the prebiotic market is the rising number of consumers seeking natural and functional foods. People are becoming more conscious of what they eat and are actively looking for products that offer health benefits beyond basic nutrition. Prebiotic formulations fit this demand perfectly, as they provide a natural and effective way to support gut health.
Furthermore, the increasing prevalence of digestive disorders such as irritable bowel syndrome (IBS), inflammatory bowel disease (IBD), and constipation has also contributed to the growth of the prebiotic market. These conditions can greatly impact a person’s quality of life, and many individuals are turning to prebiotic formulations as a way to alleviate symptoms and improve their gut health.
The COVID-19 pandemic has also played a role in the increased demand for prebiotic formulations. Research has shown that a healthy gut microbiome is crucial for a strong immune system. As people become more aware of this connection, they are actively seeking ways to support their immune health. Prebiotics, with their ability to promote the growth of beneficial bacteria, are seen as a natural and effective way to strengthen the immune system.
The market for prebiotic formulations is not limited to just food and beverages. It has expanded to include a wide range of products, including dietary supplements, infant formula, and pet food. This diversification of the market has further fueled its growth, as it allows consumers to incorporate prebiotics into their daily routine in various ways.
In conclusion, the market for prebiotic formulations is experiencing significant growth due to increasing consumer interest in natural and functional foods, rising awareness about gut health, and the need for immune support. As more research is conducted on the benefits of prebiotics, and as consumers continue to prioritize their health and well-being, the demand for prebiotic formulations is expected to continue its upward trajectory.
The Ohio State Innovation Foundation invention works as follows
Compositions containing a biocompatible sphere, biofilm-producing probiotic bacteria, prebiotic and/or prebiofilmic are provided herein. The compositions, as well as methods of preparing them and formulating them and treating or preventing disease with the compositions, are described.
Background for Prebiotic formulations
The use of probiotics to treat enteric diseases is gaining popularity. Probiotics (2001), which are becoming more popular as a treatment for enteric diseases, (Hemarajata & Versalovic (2013) Effects Of Probiotics On Gut Microbiota : Mechanisms Of Intestinal Immunomodulation And Neuromodulation Therap Adv. Gastroenterol 6:39-51)
Under the right conditions, probiotics are able to effectively prevent pathogens colonization through direct mechanisms (e.g. production of antimicrobial defences) or indirect mechanisms (e.g. stimulation of host defenses). Few probiotics species can both limit excessive inflammation and prevent pathogen invasion. It is important to note that excessive inflammation of the colon in response to colonic infections can cause protracted illness such as post-infectious IBS. The development of probiotics which are able prevent excessive immune reactions to colonic pathogens while maintaining anti-bacterial resistance would be able to help prevent both the short-term as well as the longer-term effects of enteric infections. This disclosure contains formulations that meet this unmet need, and also provide related benefits.
Aspects and embodiments” of this technology combine health benefits of probiotics with prebiotics to stimulate the exclusive growth and, in an aspect, provide bacteria in the form a biofilm onto a biocompatible sphere. The applicants have found that using a biofilm in a microsphere increases the efficacy of the treatment and the duration. The ability of probiotic biofilms to grow on surfaces to introduce bacteria to the site where wounds are present has been demonstrated. This is in conjunction with a formulation consisting of a plaster or dressing that contains a hydrocolloid, which is a gelatin-like substance, to treat wounds. There is a need to reduce probiotic doses, increase efficacy and formulate probiotic bacteria. “The compositions and methods disclosed herein address this unmet requirement and, to best of the Applicants’ knowledge have never been disclosed before.
This technology provides formulation methods that enhance the effectiveness and durability of probiotic strains introduced at a site. This technology bypasses biofilm formation, which is a rate-limiting step. This technology can be used for gut health, as well as any other area where probiotics are needed, such as the gastrointestinal tract and wound healing.
In the context of gastrointestinal health specifically and the environment in general, probiotics are a natural way to protect and restore gut microbiota to a healthy state. Unfortunately, even under optimal conditions, probiotic bacteria (as typically delivered) fail to establish, or sufficiently persist, minimizing the magnitude and duration of their healthful effects. One of the rate limiting steps is the capacity of introduced bacteria to form a lasting biofilm. When bacteria are already in the form of a biofilm (a surface adhered community) as opposed to planktonic (free-living), they more readily establish and persist. The positive effects of probiotic bacteria can be enhanced by providing them in a biofilm state; this can readily be accomplished by growing the bacteria on the surface of a biocompatible and non-toxic microsphere and associated with a biofilm. Biocompatible microspheres can be biodegradable polymers, non-biodegradable polymers, a metal, or a combination thereof. When this surface is in the form of a microsphere, prebiotic and/or prebiofilmic substances can be added as cargo to facilitate establishment and maintenance of the probiotic bacterial biofilm.
The biofilm generating probiotic bacteria adheres to surface of the biocompatible sphere and creates a film. The biocompatible microsphere can be semi-permeable, porous or solid with a hollow or solid core. The biocompatible microsphere can be hollow and carry nutritional supplements for probiotic bacteria. This is done by diffusion through the lumen. In addition, the microsphere may carry a drug or compound that is selectively against a pathogen. This agent or compound could compete with the health-inducing bacteria in the composition. In another aspect, the microsphere may contain chemical reductants or molecules or surfaces which promote adsorption in the core of the microsphere or on its surface and/or molecules or surfaces which promote absorption in the core of the Microsphere or on its surface. A novel probiotic formulation may also include a prebiofilmic in addition to the biocompatible microsphere and biofilm-generating prebiotic. The prebiofilmic can be a DNA binding protein, DNABII protein, or fragment thereof, that promotes biofilm durability and/or formation. Prebiotics are released from the hollow core to adhere to bacteria. The surface of the hollow core microsphere is porous, or semi-permeable. This allows the prebiotic to diffuse out or slowly degrade the microsphere causing leaks. The release of prebiotics from the hollow core is regulated by changing the microsphere size. Smaller microspheres will release more quickly.
Microspheres are a valuable addition to the food supplementation industry because they can provide a diffusible prebiotic cargo (nutritional supplements specific/exclusive for probiotic bacteria), which helps promote probiotic bacterial survival and establishment while limiting pathogenic challenges. The biofilm form is more advantageous for the probiotic bacterium Lactobacillus reuteri to establish in the gut than the planktonic version. L. reuteri biofilms in mice have a stronger and more durable prophylactic action against the enteropathogenic bacterium Citrobacter roentium than L. reuteri planktonic. These results lead to highly integrated examples that result in novel formulations of Probiotics with greater and longer lasting effects against dysbiosis, preventing or treating gut pathogenesis without requiring patient compliance.
In view of the advantages described above, the present invention provides a composition that comprises, or alternately consists essentially of or yet further consists of a biocompatible sphere, a probiotic bacteria capable of forming biofilms, and a prebiotic. The prebiotic may comprise, or alternatively essentially consist of or yet further consists of a nutritional complement for the probiotic bacteria. The composition may also include, or alternatively consist essentially of or still further comprise, a carrier such as a pharmaceutically-acceptable carrier or biocompatible scaffold.
The compositions can be used in vivo, or ex vivo. The compositions can be used in vivo orally, vaginally or nasally (inhalation), or intravenously (injectable), or topically as a suppository or spray (aerosol application), or dryly by mixing in soil. In one aspect, the compositions are in a dosage format. “Suitable dosage forms are, but not limited to, the following: a suppository; a powder; a liquid; a capsule; a chewable or swallowable tablet (or buccal tablet); a troche or soft chew.
In a further aspect, the method further comprises, or alternatively consists essentially of or yet further consists of, admixing an effective amount of one or more of: a nutritional supplement for the probiotic bacteria, a drug active against a pathogen or invertebrate, or a chemical reductant and/or molecule that promote adsorption (in the core or on the surface of the microsphere) and/or molecules that promote absorption (in the core or on the surface of the In another aspect, the method comprises, or alternately consists essentially, or yet further consists, of admixing a sufficient amount of one of or more: a nutritional complement for the probiotic bacteria, a drug that is active against a parasite or invertebrate or a chemical reducetant and/or molecular that promotes adsorption and/or molecules which promote absorption.
The compositions disclosed herein can also be used for therapeutic, industrial and agricultural purposes.
In some embodiments, the kit comprises, or alternatively consists essentially of or yet consists of, a composition described herein, and instructions on how to use it diagnostically, industrially or in agriculture.
It is important to understand that the invention is not restricted to any particular embodiments, since they can, of course vary. The terminology used in this document is only intended to describe particular embodiments and not to limit the invention, as the appended claims will be the only limitation.
All technical and scientific terms are defined to be understood by a person of ordinary knowledge in the field that this invention belongs. The present invention can be tested or practiced using any method or material similar to or equivalent to the ones described here. However, the preferred devices, methods and materials will now be described. The entire content of all technical and patent publications that are cited in this document is incorporated by reference. “Nothing herein should be construed to mean that the disclosure is not entitled to predate this invention by virtue of prior discovery.
Unless otherwise specified, the practice of the current technology will use conventional techniques in tissue culture, immunology and molecular, microbiology cell biology, and recombinant, DNA which are within the scope of the artist’s skill. Sambrook and Russell, eds. Molecular Cloning, A Laboratory Manual (3rd Edition), Ausubel et al. eds. (2007) Current Protocols in Molecular Biology, the series Methods in Enzymology, (Academic Press, Inc., N.Y.); MacPherson et. al. MacPherson et. al. (1991) PCR 1 : A Practical Approach, (IRL Press and Oxford University Press); MacPherson et. al. Harlow and Lane eds. (1995) PCR 2 A Practical Approach. Freshney, (2005) Culture of Animal Cells, Fifth Edition, Gait Ed. (1984) Oligonucleotide Synthesis; U.S. Pat. No. No. Hames, Higgins and Anderson eds. (1984) Nucleic Acid Hybridization. Anderson (1999) Nucleic Acid Hybridization. (1984), Transcription and translation; Immobilized Cells, Enzymes and IRL Press (1986); Perbal (1984), A Practical Guide to Molecular Cloning. Miller and Calos eds. Makrides, ed. (1987), Gene Transfer Vectors For Mammalian Cells. Cold Spring Harbor Laboratory. 2003 Gene Transfer and Expression of Mammalian Cells, Mayer and Walker editors. (1987) Immunochemical Methods in Cell and Molecular Biology, Academic Press, London; and Herzenberg et. al. Weir?s Handbook of Experimental Immunology (1996).
All numerical designations (e.g. pH, temperature and time, concentration, molecular mass, ranges) are approximates that can be changed (+) or(?) By increments of 1, or 0, as appropriate. Or alternatively, by a variation between +/- 15%, or alternatively, 10%, or alternately, 5% or alternatively, 2%. Although not always stated explicitly, it is understood that all numeral designations are preceded with the word?about?. It is also to be understood that, even though not always stated explicitly, the reagents listed here are only exemplary, and that there are equivalents in the art.
The singular form “a” or “an” is used to describe the claim and specification. The singular form?a?,?an? Plural references are acceptable unless it is clear that the context requires otherwise. The term “a bacterium” is an example. “A bacterium” includes a number of bacteria and mixtures thereof.
The term “comprising” is used in this context to mean that the compositions and methods include all elements, but do not exclude others. The term “comprising” is used to indicate that the compositions or methods do not exclude other elements but include those cited. “Consisting essentially” When used to describe compositions or methods, it means excluding any other elements that are of essential importance to the combination intended for use. A composition consisting of the defined elements would not exclude trace contaminants and pharmaceutically acceptable carriers such as phosphate-buffer saline and preservatives. ?Consisting of? This shall include excluding all ingredients other than trace amounts and any substantial steps in the method of administering compositions of the invention. The scope of the invention includes embodiments defined by these transition terms.
A ?biofilm? A biofilm is a thin layer of microorganisms or a community that can sometimes adhere to a surface, which may be inorganic or organic, along with the polymers they secrete or release, such as DNA. Biofilms are resistant to antimicrobials and microbiotics. They cause caries, periodontal disease (also known as plaque periodontal) and other diseases by living on the gingival tissue, teeth and restorations. Also, they can cause middle ear chronic infections. Biofilms may also develop on the surfaces of dental implants and stents. They can even form on contact lenses, catheters, and contact lens lines. They can grow on pacemakers and heart valves. Biofilms are estimated to be responsible for over 65% hospital-acquired infections by the Centers for Disease Control. Biofilms fungi are also a common contaminant of medical devices. They can cause chronic vaginal infection and even lead to systemic infections that are life-threatening in those with compromised immune systems. Biofilms are also involved in many diseases. “For example, Pseudomonas infections in cystic fibrosis can lead to antibiotic-resistant biofilms.
A ?prebiotic? “A?prebiotic? Prebiotics, such as oligosaccharides are food ingredients that are not digestible by the subject (e.g. a mammal like a human) and which stimulate or inhibit the growth of beneficial bacteria. Prebiotics may stimulate the growth or activity of a small number of bacteria within the subject.
A ?prebiofilmic? “A?prebiofilmic?
Click here to view the patent on Google Patents.