Invented by Edward T. Maggio, Aegis Therapeutics LLC
One of the key drivers of the market for compositions for drug administration is the increasing prevalence of chronic diseases such as diabetes, cancer, and cardiovascular diseases. These diseases require long-term treatment with drugs, and the availability of different compositions for drug administration allows patients to choose the most convenient and effective method of drug delivery. For example, patients with diabetes may prefer to use insulin pens or pumps rather than traditional injections, while cancer patients may benefit from targeted drug delivery using nanoparticles.
Another factor driving the market for compositions for drug administration is the growing demand for personalized medicine. Personalized medicine involves tailoring drug treatments to individual patients based on their genetic makeup, lifestyle, and other factors. Compositions for drug administration play a crucial role in personalized medicine, as they allow drugs to be delivered in specific doses and at specific times to optimize their therapeutic effects.
The market for compositions for drug administration is also being driven by advances in drug delivery technology. New technologies such as nanotechnology, microfluidics, and 3D printing are enabling the development of novel drug delivery systems that can improve drug efficacy, reduce side effects, and enhance patient compliance. For example, 3D printing can be used to create personalized drug delivery devices that are tailored to individual patients’ needs.
Despite the many opportunities in the market for compositions for drug administration, there are also challenges that must be addressed. One of the biggest challenges is ensuring the safety and efficacy of these compositions. Different compositions may have different pharmacokinetic and pharmacodynamic properties, and it is important to ensure that they are properly tested and validated before they are used in clinical settings.
Another challenge is the high cost of developing and manufacturing compositions for drug administration. Developing new drug delivery systems can be a complex and expensive process, and manufacturers must balance the need for innovation with the need to keep costs under control.
In conclusion, the market for compositions for drug administration is a rapidly growing and dynamic field that is driven by a range of factors including the increasing prevalence of chronic diseases, the demand for personalized medicine, and advances in drug delivery technology. While there are challenges that must be addressed, the potential benefits of these compositions for patients and healthcare providers are significant, and the market is likely to continue to grow in the coming years.
The Aegis Therapeutics LLC invention works as follows
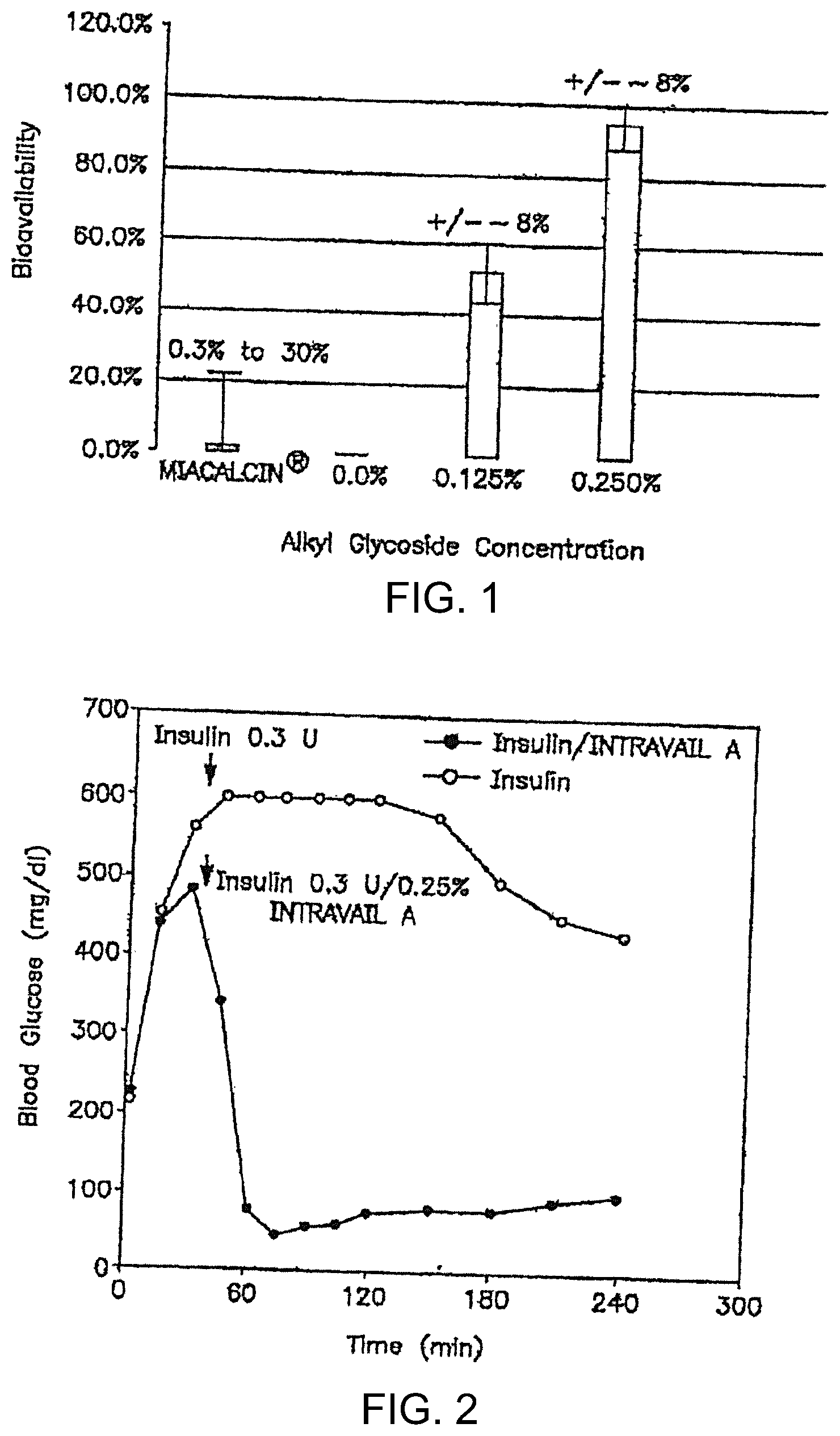
The present invention provides compositions, methods, and means for increasing the bioavailability and effectiveness of therapeutic agents in a subject.” Compositions may include at least one alkylglycoside and at most one therapeutic agent. The alkylglycoside’s length is approximately 10 to 16 carbon atoms.
Background for Compositions for drug administration.
Field of Invention
The present invention is generally applicable to compositions that contain a pharmaceutically active ingredient or an alkylsaccharide.
Background Information
Many therapeutic agents combine with different surfactants. Surfactants can be irritating to skin and other tissues. Surfactants can also denature proteins, thereby reducing their biological activity. The ability to safely deliver such agents to their intended sites of action is another limitation in the development and application of these agents. The ideal surfactant for therapeutic agents will be stable, non-toxic, non-irritable to skin and mucosal surfaces, and have antibacterial activity. It will also enhance the passage of therapeutic agents through membrane barriers, without affecting the structural integrity or biological function of the membrane, and increase the bioavailability.
Despite the many benefits of peptides or proteins as therapeutic agents, they are not suitable for oral administration. Typically, injections are required. This is a serious problem. Peptides are significantly less stable than small-molecule drugs. To avoid undesirable degradation, peptides must be stored and prepared with care. Immunogenicity can occur with certain proteins, especially proteins that contain substantially non-naturally occurring sequences of amino acids. Immune cells can be recruited to the injection site and an immune response, either humoral or cell-mediated, may be inducible. It is known that aggregation of peptides is more likely to trigger an immunegenic response than monomers. This can be avoided in a more or less severe manner if the peptide is absorbed directly from the gastrointestinal system into the systemic circulation. Although there are many clinical uses for therapeutic proteins and/or peptides, the actual number of therapeutic proteins and/or peptides on the market today is very small in comparison to the amount of pharmaceuticals that have been chemically synthesized orally active. A large number of alkylsaccharide delivery enhance agents have been developed in recent years. These include molecules that offer intranasal bioavailabilities comparable to injections. Recent developments in intranasal delivery of proteins and peptides offer new clinical applications for peptides and proteins. However, few if any peptides can be administered orally due to low oral bioavailability. Numerous studies have been done to prove oral bioavailability of various peptide drugs. The studies involved a range of absorption enhancers and physical processes like micronization. Insulin, for example, was the only formulation specifically designed for oral delivery. It showed a 3% oral bioavailability according to Babwin et. al., 2009. Calcitonin only showed a 0.5-1.4% oral bioavailability. (Bucklin 2002). Parathyroid hormone was found to have 2.1% oral bioavailability according to Leone-Bay and colleagues, 2001. Two main biochemical issues limit oral absorption of Peptides. The first is the susceptibility to hydrolysis of peptides in the gastrointestinal tract. The second is due to poor intestinal absorption across the mucosal membrane.
It has been demonstrated that some peptides can be hydrolysed or slow-metabolized by incorporating non-standard amino acid sequences. This allows drugs to stay active for longer periods of time, as they can be incorporated with non-standard aminoacyl residues. Non-standard amino acid are amino acids that do not belong to the 22 L-amino acid naturally found in proteins. Non-standard amino acids can be used in either the D or the L configuration. A few examples include, but are not limited to, allylglycine, (2S,3R,4S)-?-(carboxycyclopropyl)glycine, ?-cyclohexylglycine, C-propargylglycine, ?-neopentylglycine, ?-cyclopropylglycine, N-lauroylsarcosine sodium salt, N-(4-hydroxyphenyl)glycine, N-(2-furoyl)glycine, naphthylglycine, phenylglycine, lanthionine, 2-aminoisobutyric acid, dehydroalanine, gamma-aminobutyric acid.
Some specific examples of non standard amino acids used in drugs include D-4-hydroxyphenylglycine which is incorporated into the antibacterial drug Amoxicillin, D-phenylglycine which is incorporated into the antihypertensive drug Enalapril, and (2R,3S)-phenylisoserine which is incorporated into the antineoplastic drug Taxol.
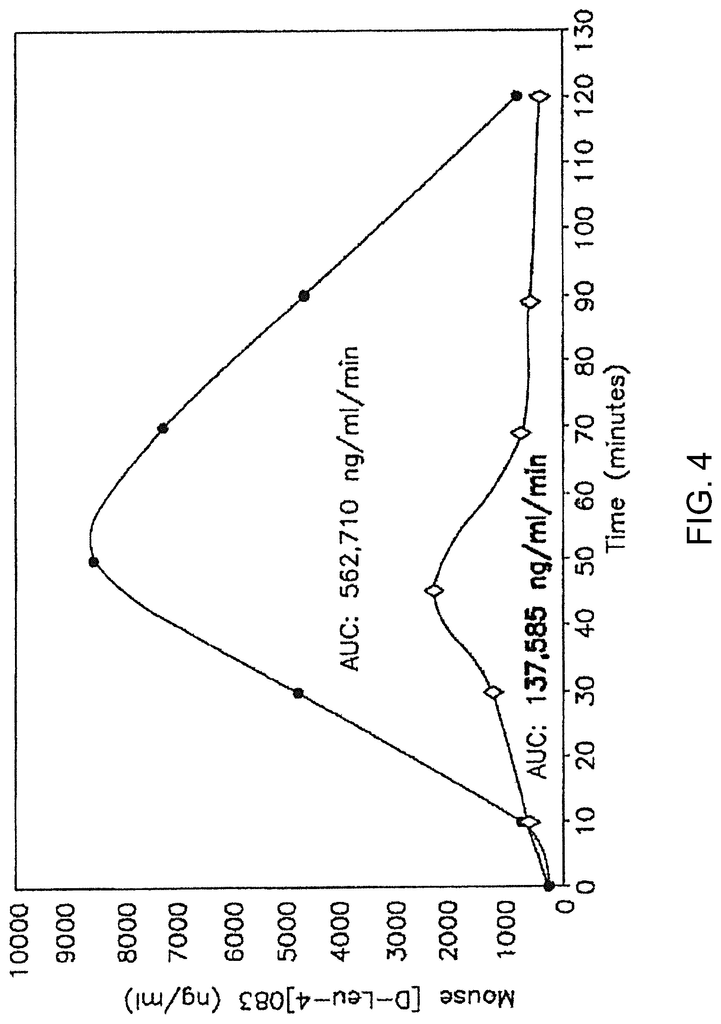
D-2-Naphthylalanine, a peptide drug, is added to the Nafarelin endometriosis medication. To increase the stability of peptide drug, D-isomers are often used of naturally occurring L’amino acids. D-amino acids stabilized peptides include, among others, the anti-obesity peptide D?Leu-OB3 by Lee et. al. (2010) and the CCR5 anti HIV drug D?ala-peptide (DAPTA), by Ruff et. al. 2001.
Enzymatic hydrolysis can also be reduced or eliminated through the addition of specific enzyme inhibitors, such as amastatin (bacitracin), bestatin (boroleucin), borovaline and aprotinin) and trypsin inhibit (among others).
When presented in aqueous solutions, the alkylsaccharides and small molecules or peptides are able to increase oral absorption. These solutions have a higher concentration of alkylsaccharides than the critical micelle concentration (CMC). An example includes the oral delivery of octreotide in aqueous solution using dodecyl-beta-D-maltoside as the alkylsaccharide absorption enhancer. Oral delivery of a synthetic insulin sensitizer leptin-related using an aqueous solution of dodecylmaltoside. Yet another example is oral delivery of exenatide and pramlintide using dodecyl-beta-D-maltoside in an aqueous solution as the alkylsaccharide absorption enhancer. Another example is oral delivery of heparin with tetradecyl-beta-D-maltoside in an aqueous solution to enhance the alkylsaccharide absorption (see Maggio and Grasso; Novakovic, et. al.., Peptides 43 (2013) 167-163); Leinung M. C. et. al. Regulatory Peptides 179.33-38 (2012); Yang, et. al.
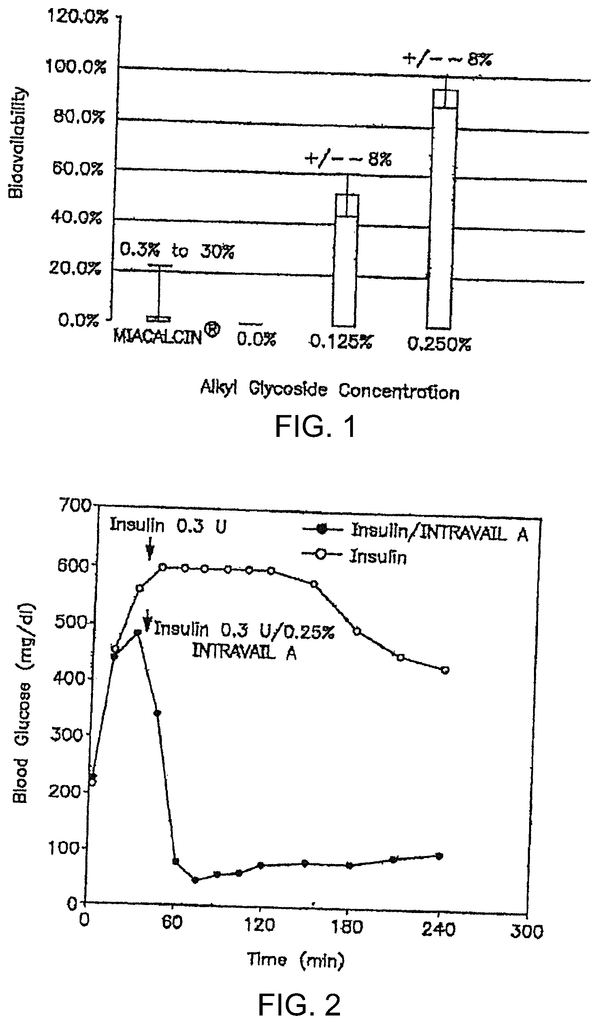
The present invention focuses in part on the development and use of a therapeutic composition that contains a drug enhancing ingredient. This agent is useful for increasing drug absorption and bioavailability while avoiding adverse toxic effects. The invention’s drug enhancing agents contain a nontoxic surfactant that contains at least one alkyl glycoside or saccharide alkyl ester. The therapeutic compositions of invention have the advantage of allowing administration and delivery at high bioavailabilities, even at low concentrations. (their NOAEL’s). The present invention includes compositions that include alkyl glycosides or saccharide alkyl esters and a therapeutic agent (e.g. small molecule organic drugs molecules, low molecularweight peptides like Exenatide, GLP-1, and other such therapeutic agents), methods for administering and using compositions (e.g. via the oral, nasal, nasolacrimal or pulmonary route and methods to ameliorate a disease condition in a subject through administration of such compositions
Previously, there were no examples of alkylsaccharides being used as absorption enhancers in tablets or solid dosage forms. Dodecyl Maltoside and n-tetradecyl Maltoside are alkylsaccharides that have a high solubility in water. However, they dissolve slowly because large amounts of their molecules contain hydrophobic linear alkyl chain. To ensure that these alkylsaccharides are completely dissolved in aqueous solutions, they should be heated gently and the container containing the aqueous media is stirred or agitated for between 15 and 30 minutes. To ensure that the alkylsaccharide is completely dissolved, it should be heated gently. It was therefore not expected that solid dosage forms containing an Alkylsaccharide or the drug substance would be able to benefit from the absorption-enhancing effect of alkylsaccharide. The slow dissolution in the aqueous contents of the alkylsaccharide would produce a sufficient high concentration or comparable concentration to the one achieved in the aqueous contents before the drug substance. Furthermore, the molecules of alkylsaccharide diffuse away from one another throughout the gastrointestinal contents, reducing the relative concentrations. Surprisingly solid dosage forms containing an alkylsaccharide and a pharmacologically activated substance along with other inactive excipients were found to increase oral bioavailability. Other inactive excipients that may be added to the active pharmaceutical substance or alkylsaccharide absorb enhancer include hypromellose and magnesium stearate as well as microcrystalline cellulose and polyethylene glycol.
The present invention is a surfactant composition that contains at least one alkyl sugarside and/or at most one saccharide alkyl ester. When mixed, blended, or admixed with a therapeutic agent or biologically active compound, this surfactant stabilizes biological activity and increases bioavailability.
The invention also includes a method for administering a therapeutic composition that contains a surfactant, which may include at least one alkyl sugarside and/or saccharidealkyl ester. The surfactant is used to increase the stability and bioavailability the therapeutic agent.
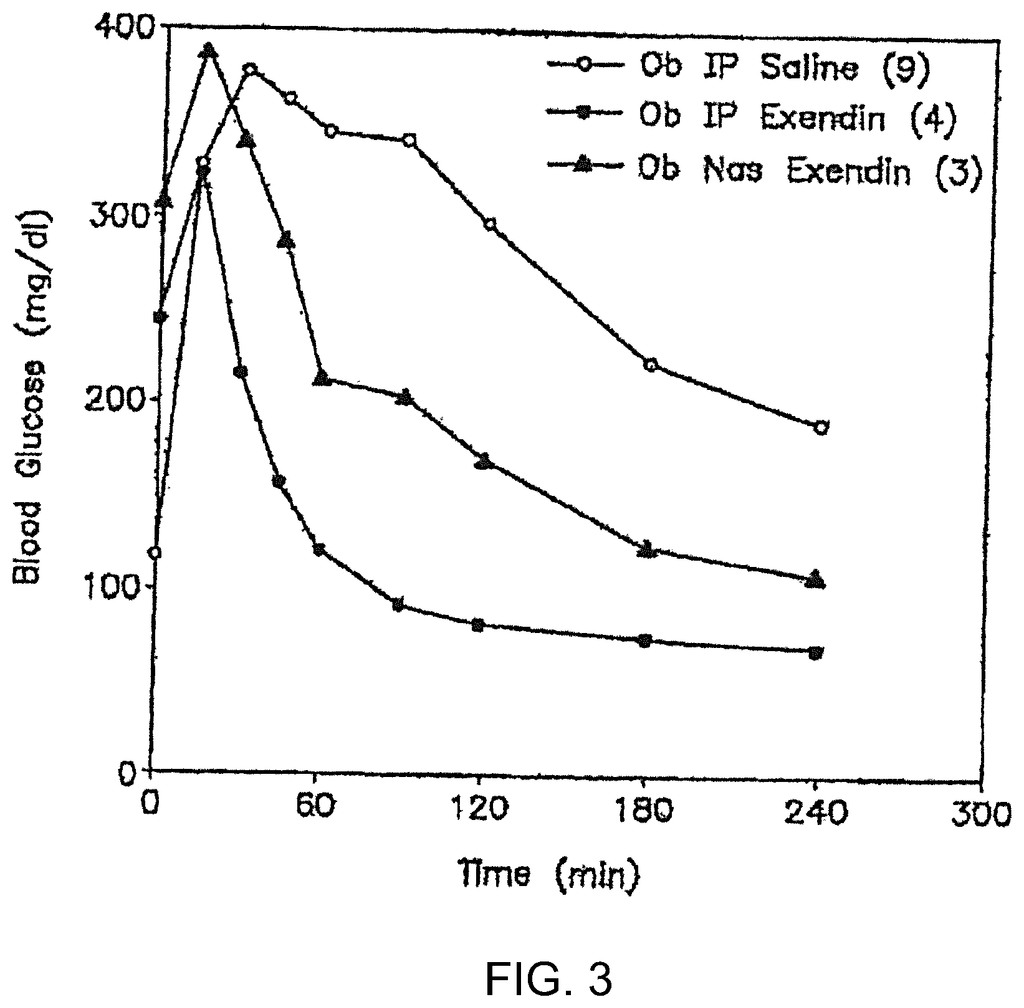
Another aspect of the invention is that it provides a method for increasing the absorption of low molecular weight compounds into the circulatory systems of subjects by administering them via the oral, nasal, nasolacrimal or pulmonary routes. The surfactant can be a nontoxic, nonionic hydrophobic alkyl linked by a linkage with a hydrophilic Saccharide. These low molecular weight compounds include, but are not limited, nicotine, interferon PYY, GLP-1 and synthetic exendin-4. Antisense oligonucleotides and interfering RNA molecules are also low molecular-weight compounds (e.g. siRNA or RNAi).
The present invention also provides a method for treating diabetes. This includes administering insulin to the subject via the nasal, buccal, or oral cavities.
The present invention also offers a method for treating congestive cardiac failure in a subject. This includes administering to the subject via the oral, nasal, or nasolacrimal delivery route, a therapeutically efficacious amount of a composition consisting of a GLP-1 protein or functional equivalent thereof and an absorption increasing quantity of a nontoxic, nonionic glycoside with a hydrophobic hydrophobic linkage to a waterphilic saccharide.
Another aspect of the invention is a method for treating obesity and diabetes in a subject. It involves administering to the subject via the oral or ocular nasal, nasolacrimal or CSF delivery routes, a therapeutically-effective amount of a composition consisting of a PYY protein or functional equivalent thereof and an absorb increasing amount of a nontoxic, nonionic glycoside with a hydrophobic linkage to a waterphilic saccharide. This will treat the subject.
Another aspect of the invention is a method for increasing the absorption of low molecular weight therapeutic compounds into the circulatory systems of subjects by administering the compound via the oral or ocular nasal, inhalation, or CSF delivery route. The compound has a hydrophobic hydrophobic alkyl chain joined by a linkage with a hydrophilic Saccharide.
The present invention also allows for increased absorption of low molecular weight therapeutic compounds into the circulatory systems of subjects by administering the compound via the oral or ocular nasal, nasolacrimal or pulmonary routes.
Click here to view the patent on Google Patents.